 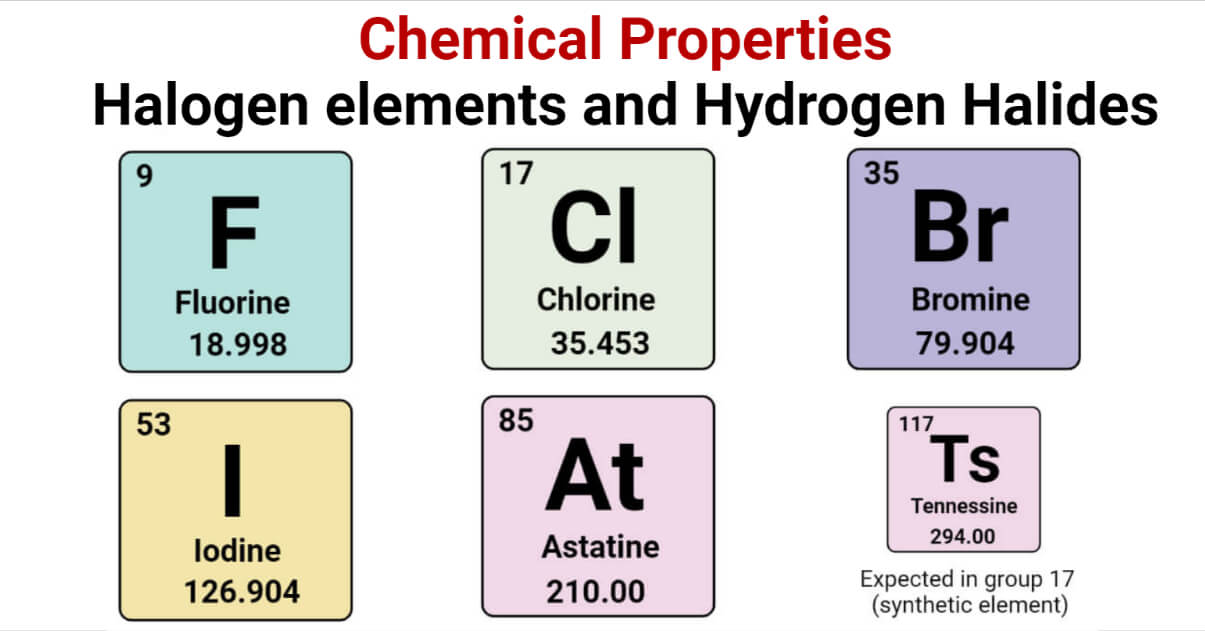 
These high reactive nonmetals exhibit colour and. As a result, the halogen group is the only elemental group that, at ambient temperature and pressure, includes all three of the usual states of matter. Halogens elements belong to Group 7 on the periodic table and exhibit unique chemical and physical properties. Tennessee is thought to be solid by scientists. Many of the simple chalcogen halides are commonly used reagents. These are known as chalcohalides or chalcogen halides. You need to know details about chlorine, bromine and iodine. They are called the halogens because they react with metals to form salts (from Greek hal meaning ‘salt’ and gen meaning ‘to produce’). They create brittle solids and have poor heat and electrical conductivity. Chalcogens also commonly form compounds with their neighbor group, the Halogens (group 17). The group 7 elements are placed on the right of the periodic table. Group VII, often known as group 7, or group 17 in more recent IUPAC terminology, designates the halogens. Halogens are located on the periodic table's right side, just to the left of the noble gas group. They belong to a group of gases that were historically utilised as aerosol propellants and in refrigeration. They include Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and Radon (Rn). It is utilised in the metallurgical processing of iron and steel.ĬFCs, or chlorofluorocarbons. Elements that are in Group 18 (VIIIa) of the periodic table are called noble gases. Because of its photosensitivity, it is employed in photography.įluoride in calcium (CaF 2 ). It is used to season meals and is edible. Some well-known halogen-containing compounds include:Ĭommon salt or table salt (NaCl). In the meanwhile, fluorine is utilised to create Teflon resins, lubricants, and antifreeze.Įxamples of Everyday Substances with HalogensĪerosols once included the halogen-containing chemical CFC. Iodine has antibacterial halogen properties. The term halogen means salt-former and compounds containing. The photography business uses brominated chemicals (bromides) as a material and as sedatives. The halogens are five non-metallic elements found in group 17 of the periodic table. Nuclear medicine uses the isotopes of astatine and iodine. There are six halogens- fluorine, chlorine, bromine, iodine, astatine, and. In pharmaceuticals, halogens are employed. Halogens can be defined as elements that form salts when they react with metals. 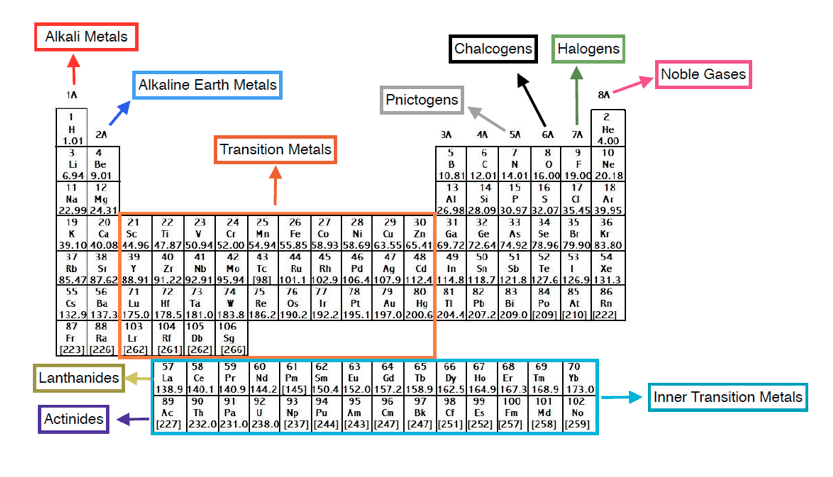
Halogen lamps use iodine and bromine to provide a brighter glow than ordinary incandescent lights. halogensGroup 17 (or VII) in the periodic table consisting of fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Both of these substances are frequently employed as flame retardants. 
To cure wounds, sanitise surfaces, and safeguard swimming pools and spas, disinfectants like chlorine and bromine are utilised. Halogens are a group of non-metal elements in the periodic table, consisting of fluorine, chlorine, bromine, iodine, astatine, and tennessine. The uses of halogen elements are numerous. The term 'family' is synonymous with the term 'group'. Chemical families tend to be associated with the vertical columns on the periodic table. The decreasing order of electronegativity F>Cl>Br>I is what is the order of electronegativity among halogens. In chemistry, a family is a group of elements with similar chemical properties. The most electronegative element is fluorine. Halogens are a group of elements on the periodic table found in group 17. On the Pauling scale, its electronegativity is 2.5 as well (or less). They are all oxidising substances as a result. The elements with properties intermediate between those of metals and nonmetals are called metalloids (or semi-metals).Halogens create monovalent ions (-1), meaning they lack the last electron needed to complete their energy level. \), metals occupy the left three-fourths of the periodic table, while nonmetals (except for hydrogen) are clustered in the upper right-hand corner of the periodic table. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
